We use water for transfer of heat because it absobs a lot of heat. (evaporation heat: 540 cal/gr) is inexpencive easly pumped.
Water are required for cooling of product and equipments.
Water is the universal solvent. All Water contains dissolved gases (02, Co,) and chemicals (Cations like Ca, Mg, Na, Fe and Anions like CO. HCO3 CL, SO4) and suspenden solids, bacteria

OBJECTİVES OF ADVANCED WATER TREATMENT TECHNOLOGY
Keeping heat transfer surfaces clean, more efficient heat transfering reduce energy and maintenence cost.
Prevevent Corrosion
Extend the life of systems and reduce maintenance cost
Incomplate heat transfer, insufficient cooling or heating can reduce product quality. production efficiency.
İncreasing quality and reducing production cost
Protect worker and human health
Protect the Evironment
This impurities causes following major problems for generation of steam;
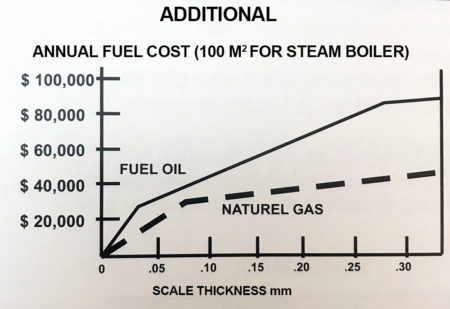
- SCALE scale, reduces heat transfer increases energy consuption. In boiler overheating-tubes metal fatigue causes of pipes puncures Cooling rates reduce
- Sludge and Fouling cuses blokages.
- Corrosion is electro-chemical reaction and cause metal loss pipe puncutre. Reasons of corrosion are dissolved oxgen, acidic water, electrical potential diffrences. Corrosion produce Metal oxides sludges useful life of system is reduced, maintanence cost is increased.
- Steam contains impurities.
- Microbiological reproduction
Around in cooling towers splashed water particules, when inhaled, cause disease. Examle; legionary disease
The elimination of problems described above is Water treatment.
WATER TREATMENT IN INDUSTRY
Pre-Treatment
- Filtration: Suspended solids are removed by filtration.
- Softening of water: Calcium and Magnesium ions gives Hardness. Calcium and Magnesium ions are exchaned for Sodium ions by Cationic resins. Solublity of Sodium salts are very high and can not form scale.
2R-Na + CaCO3 R-Ca + Na2CO3
If Resin washed with salt Resins back to the original state
2R-Ca + NaCL R-Na + CaCL2
- In dealkalization, the ions contributing to alkalinity (carbonate, bicarbonate, etc. are given Carbon dioxide and Carbonic asid) removed and replaced Chloride Ions by resins.
HCO3 H20 + CO,
H2O + CO2H2CO3
Dealkalization processes utilizing weak acid cation resin or strong acid cation resin in a split stream process, exchange cations with hydrogen.
Ion exchange Process does not change conductivity. but reduces chemical consumption.
- Degasification: Oxygen ve Carbon dioxside are the cause of corrosion and removed by degasser. Non-removing residue CO2 ve O, are trapped by chemicals.
TREATMENT OF FEED WATER İN BOİLER
If there is no Pre-treatment system or it does not work effectively, because of leak from preteatment, water treatment of feed water is necessary.
Factors consider:
- Chemicals and impurities concentration Control
- Structure and Pressure of Boiler
Usually steam does not contain solid material. Solid material entering with feed water accumulates in the boiler. For example,10 tons/h feed water enter the boiler producing 10 tons of steam . If feed water contains 300 ppm=300gr/ton, 300 gr/h solid materialsenter the boiler. 3kg. solids accumulate in the boiler in 10 h, 30 kg solids in 100 h.
High solids must be thrown out of the boiler (Blowdown) Thus solid concentration is kept within the limits.Blowndown is necessary but it is loss of energy.
55 kg fuel-oil spent with 1000 kg. blowndown
Water concuption increase with blowndown.
Blowdown should be kept to a minumum .
For this, Calcium and magnesium kations and solids should be kept at minumum level
- There for pretreatment is important
- Also Special polymer should be used.
- Autamatik bowndown system should be used.
- Correct chemicals should be fed at right rated according to the boiler water control parameter.
- Feed Water and boiler water should be freqıently
- Precautions should be taken according to the analysis results
- Chemicals and sevice quality is important.
USES OF POLYMERS
Three Mechanisms are effective in prevent scale formation with specials polymers :
- Crystal modification:Polymer molecules penerate smoot cube- shaped CaCO3 crystals and prevent crystallization.
- Threshold effect: They prevent scale formation if dosed below stoichometric. For example; Acrilic acid polymers with moleculer weight of 3000 are dosed 2-10ppm .
- Sequestration: Polymers makes water soluble complex in stoichiometric rate with cations of metal (Ca, Mg, Fe, Si etc).
- WTC-BPT ve WTC-C15 are devoped with polymer technology They prevent sclae formations without chemical rections reduce sediment formation and bowndown so energy loss.

SCALE FORMATION

CORRUPTION

Corrosion and Prevention
Galvanic Corrosion: Due to electro-potential diffrences.
Different metals such as Iron and Copper, conductive deposits,stratch on surface, welded spots make diffrences on electro-potential.
Dissolved oxygen : Henry Law : C=kp : p=partial pressure on solution, k= is constant. The solibility of oxygen decreases with increasing temperature. Example:in 59 F- 10ppm 176 F – 2.8 ppm, 212 F ise 0.5 ppm. According to standartlar O2<7ppb
Oxygen which can not remove in degasser,is chemically bounded.
Asidic corrosion: Low pH/alcalitnity values/ are the cause of corrosion pre-boiler auxiliary facilities and condence lines,cooler systems pH should be 8,5.
Caustic corrosion: High amont of caustic is cause of the corrosion . caustic dissolves protective magnetite layer. (Fe304)
caustic must be neutreized by disodium phosphate.
Descaling with acids: Acids dissolve magnetite layer. Inhibitors are necessary.Passivation should be made after descaling.
Dissolved Carbon dioxside: The sorce of Carbon dioxide are Carbonate and Bicarbonate anion in boiler. These break down and become carbon dioxide in boiler. 1 ppm carbonate, gives 0.35 ppm carbon diokside . Carbon diokside circulate with steam and turns carbonic acid when steam condansing. Carbonic asid is corrosive in condens lines. Carbonic acid neutralized with volatile amines, Which type of amines use, is determined by the length of condensate line.